Phosphates
Chemistry
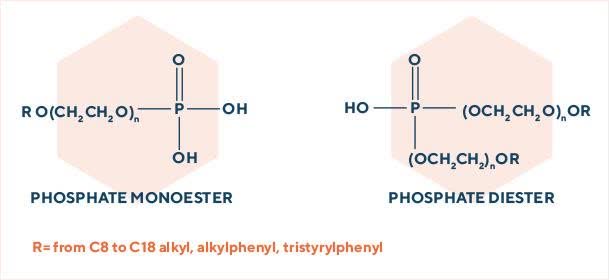
In this class of surfactants, the anionic group is linked to the molecule through an oxygen-phosphorus bond.
We manage two different phosphatation processes: with phosphoric anhydride and with polyphosphoric acid.
The starting substrate could be a fatty alcohol or an already ethoxylated intermediate (alcohol ethoxylates, alkylphenol ethoxylates, tristyrylphenol ethoxylates etc.). With both processes, a mono-ester/di-ester mixture is obtained.

We obtain the desired mono/diester ratio through careful selection of process parameters (reagent molar ratio, phosphating agent, process temperature etc.). Our phosphates can be produced in both acid and neutralized form and are stable also in highly alkaline conditions.
Our phosphate surfactants derived from an ethoxylated intermediate carry both, a polyoxyethylene chain and an anionic group so they are strong Dispersing Agents providing a double stabilization: steric and electrostatic.
The Dispersing power is further enhanced if aromatic rings are present in the hydrophobic moiety: the bulky hydrophobic structure with four aromatic rings of tristyrylphenol ethoxylate phosphates guarantees excellent absorption on the solid to be dispersed.
Applications

Due to the combination of high dispersing power and high stabilizing properties our phosphate surfactants show excellent performances in Active Ingredient Dispersions for Crop Protection Formulations and in Pigment Dispersions for the Textile and Paint sectors.
In the Emulsion Polymerization process they are used as co-surfactants:besides their contribution to the stabilization of the latex particles, they bring water and scratching resistance properties to the polymer film.
Our phosphate surfactants are used as well as corrosion inhibitor, for example in the Oil and Gas sector.
The stability in highly alkaline conditions allows them to be applied in sectors that required high pH such as in the Industrial and Institutional Cleaning.
